
Evaluation of Overall Response Rate and Progression-Free Survival as Potential Surrogate Endpoints for Overall Survival in Immunotherapy Trials | Clinical Cancer Research

Response rates Overall response rate and depth of response according to... | Download Scientific Diagram
PLOS ONE: Bevacizumab Addition in Neoadjuvant Treatment Increases the Pathological Complete Response Rates in Patients with HER-2 Negative Breast Cancer Especially Triple Negative Breast Cancer: A Meta-Analysis

ESMO 2021: A Phase 2 Prospective Trial of Frontline Cabozantinib in Metastatic Collecting Duct Renal Cell Carcinoma: The BONSAI Trial
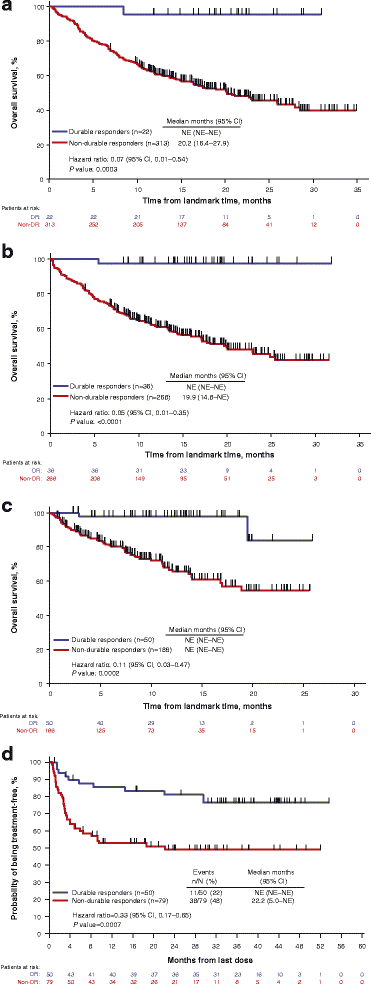
Durable response rate as an endpoint in cancer immunotherapy: insights from oncolytic virus clinical trials | Journal for ImmunoTherapy of Cancer | Full Text

Evaluation of Overall Response Rate and Progression-Free Survival as Potential Surrogate Endpoints for Overall Survival in Immunotherapy Trials | Clinical Cancer Research













![PDF] Incorporating Clinical Trial Data Into Daily Cancer Care | Semantic Scholar PDF] Incorporating Clinical Trial Data Into Daily Cancer Care | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/30f4b9adffa198bf79a177643e74ec64b4107203/3-Table1-1.png)


